The global healthcare data monetization market was valued at USD 580 million in 2025 and, according to MarketsandMarkets, is projected to reach USD 1.16 billion by 2030, growing at 14.9% per year. The fastest-growing segment is pharmaceutical and biotechnology companies, which are willing to pay for structured clinical data to support research, development, and regulatory submissions. Within this ecosystem, laboratory data (test results, biomarker trends, longitudinal patient profiles) represents one of the highest-value clinical and commercial data sources. The problem is that most laboratories producing this data cannot participate in this market.
The reason is not lack of demand. According to LLR Partners, 90% of pharmaceutical companies now have dedicated Real-World Data (RWD) and Real-World Evidence (RWE) teams, and spending in this area is growing between 8% and 10% per year. The FDA, through the 21st Century Cures Act, has formalized the use of real-world evidence to support regulatory decisions, including therapeutic indication expansion and post-market safety studies. PwC, in its analysis of the value of RWE for the pharmaceutical industry, identified that mature applications already include outcomes studies, patient cohort design, and post-approval surveillance, while emerging applications (synthetic control arms, optimization of commercial activities) promise even greater value. The demand side is ready. The supply side, at most clinical laboratories, is not. And the reason is not lack of volume. Laboratories generate millions of results per year, covering cardiac, oncological, metabolic, endocrinological, and infectious disease biomarkers. The problem is that these results, in the form they are produced, have no commercial value. They sit in PDFs, with local nomenclatures, inconsistent units, no semantic mapping, and no longitudinal traceability. A fasting glucose result issued in PDF with a single laboratory's internal nomenclature is not sellable data. It is an operational record.
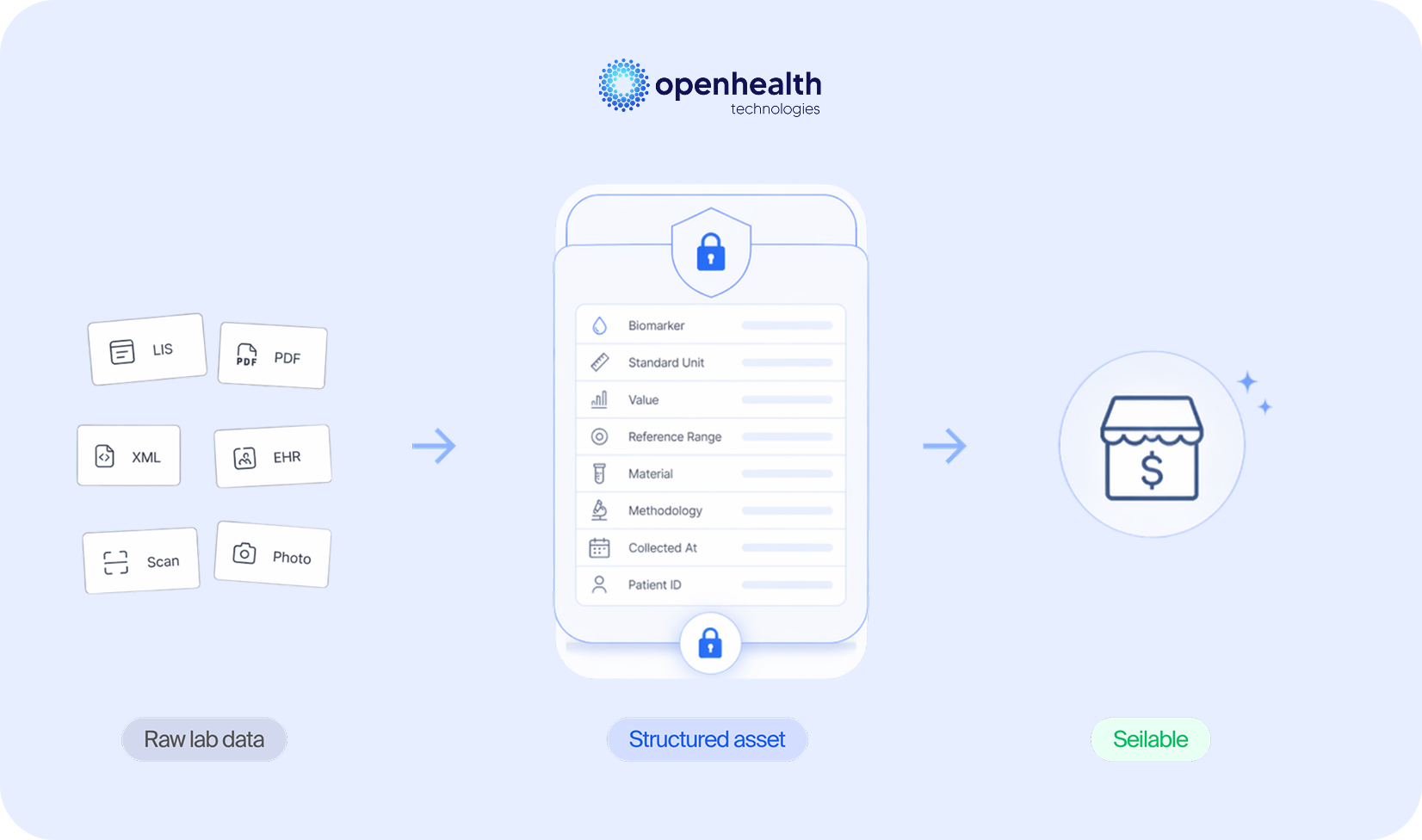
For laboratory data to have market value, it must meet four simultaneous requirements: be structured (not in document format), be semantically harmonized (standardized nomenclatures and units with LOINC mapping), be longitudinally consistent (comparable over time, even when the patient changes laboratories), and comply with privacy regulatory frameworks (LGPD, GDPR, HIPAA). Without these four layers, the data remains locked inside the operation. The potential value is concrete. Berger and Doban, writing in Clinical Chemistry and Laboratory Medicine, estimated that the informational value of integrated laboratory data corresponds to 12% to 18% of total health system operating costs. This value does not come from individual test results, but from the ability to aggregate, compare, and analyze results over time and across populations. It is the difference between a point-in-time report and a longitudinal, clinically valid dataset.
The HFMA (Healthcare Financial Management Association) defines data monetization as the use of data to create economic value, either by driving new revenue or reducing costs. The publication noted that healthcare data are strategic assets with the potential to improve both the top and bottom lines of an organization. For laboratories, this means the same data that today serves a single report can, when structured and harmonized, feed multiple value channels simultaneously. The channels are diverse. Structured lab data can feed clinical research programs (patient identification, synthetic control arms, feasibility studies), epidemiological surveillance (population-level biomarker trend monitoring), digital health platforms (healthtechs that need data as input for their products), health insurers (loss ratio management, chronic disease monitoring), and the pharmaceutical industry (real-world evidence for regulatory submissions, indication expansion, pharmacovigilance). Each channel has a distinct willingness-to-pay profile, but all depend on the same prerequisite: data that is comparable, traceable, and clinically valid.
From a competitive standpoint, this transition redefines what it means to be a clinical laboratory. A lab that limits itself to issuing reports competes on price and convenience, in a market with over 23,000 laboratories in Brazil alone (according to Pessoa and Rios in the Brazilian Journal of Health and Pharmacy). A lab that structures its data competes on value, accessing revenue channels that simply do not exist for those operating with unstructured data. The difference between the two is not analytical capability. It is data infrastructure.
This distinction becomes more relevant as the laboratory test itself becomes a commodity. When all laboratories offer equivalent analytical quality, turnaround times, and costs, the competitive difference shifts from technical execution to the ability to transform generated data into information that is integrable, actionable, and marketable. Laboratories that invest in data harmonization are not just improving internal operations. They are opening a new business model. The prerequisite for this transition is harmonization. Without a layer that transforms raw results into semantically consistent, longitudinally traceable, and clinically valid data, the laboratory remains locked in the transactional model of report issuance. With that layer, it becomes a strategic data producer capable of accessing markets currently dominated by large international networks that have already invested in this infrastructure.
It is within this context that OpenHealth Technologies is positioned. The OpenHealth Platform automatically correlates multiple data streams with rigorously validated logical layers of laboratory tests, covering over 3,500 biomarkers with native LOINC mapping. The platform ingests data in any format (including PDFs, XML, HL7, FHIR, and manual input) and delivers structured, semantically harmonized data ready for consumption by any downstream application. For laboratories, this means transforming the asset they already produce daily into a marketable product, opening revenue channels that depend not on test volume, but on the quality and structure of the information generated.
Learn how your laboratory can transform data into a new revenue stream.